Human Anti-Influenza Nucleoprotein Antibody IgG Titer Serologic Assay Kit
Catalog Number:
B20S85
Background:
Seasonal influenza (Flu) caused at least 25 million illnesses. A rapid and effective assay kit detecting the levels of anti-Flu Nucleoprotein (NP) in human serum can facilitate research on antibodies produced in response to influenza virus infection or vaccines. Based on WHO recommendation, we developed test kits for NP of 4 different strains: Influenza A seasonal H1N1, Influenza A seasonal H3N2, Influenza B seasonal Victoria Lineage and Influenza B seasonal Yamagata Lineage, see below strain info.
Applications:
This kit is developed for serologic test for human IgG titer of Anti-influenza nucleoprotein (NP) antibody in serum/plasma or purified human antibody (monoclonal & polyclonal) in vitro. It is for research use only.
Description:
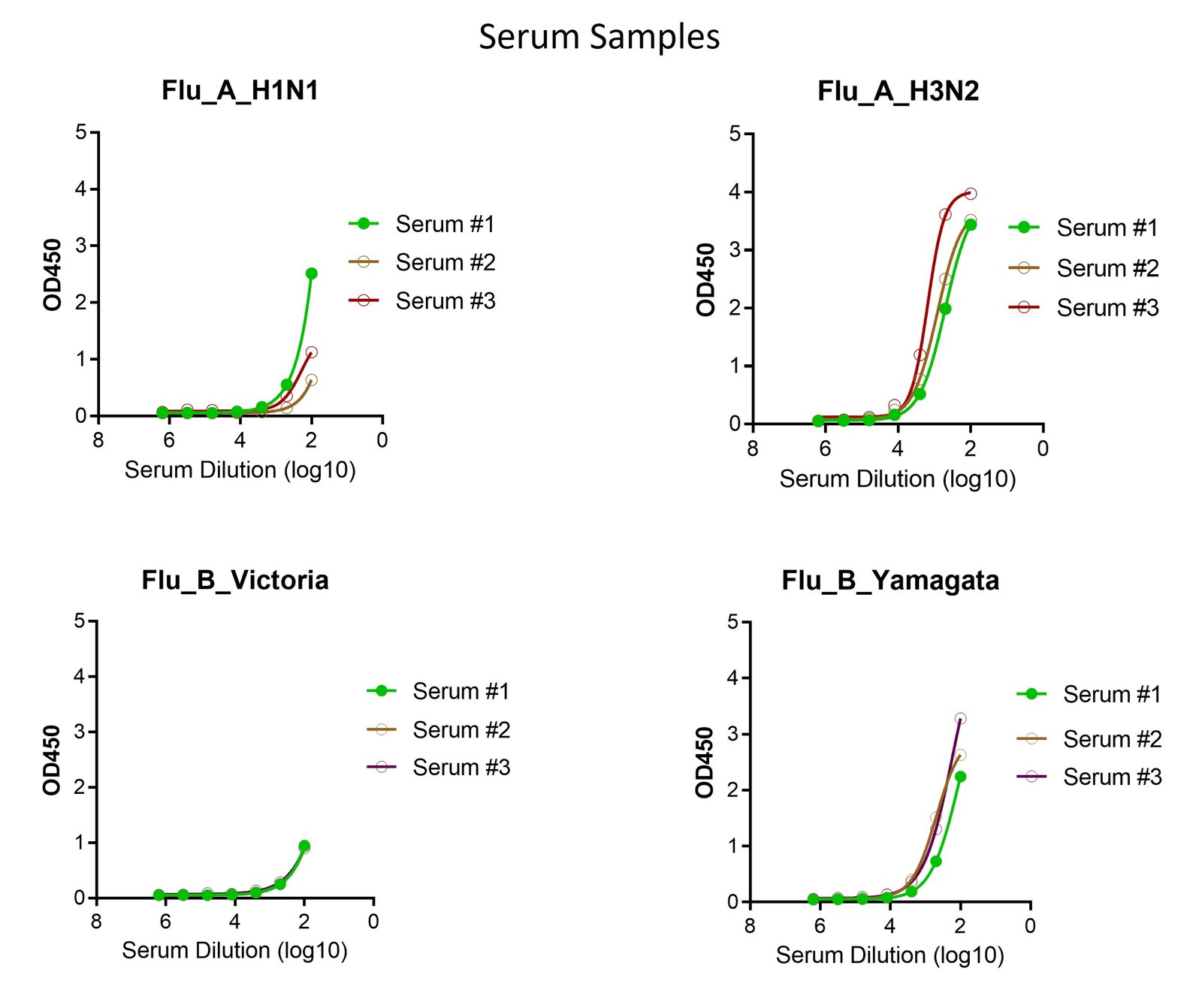
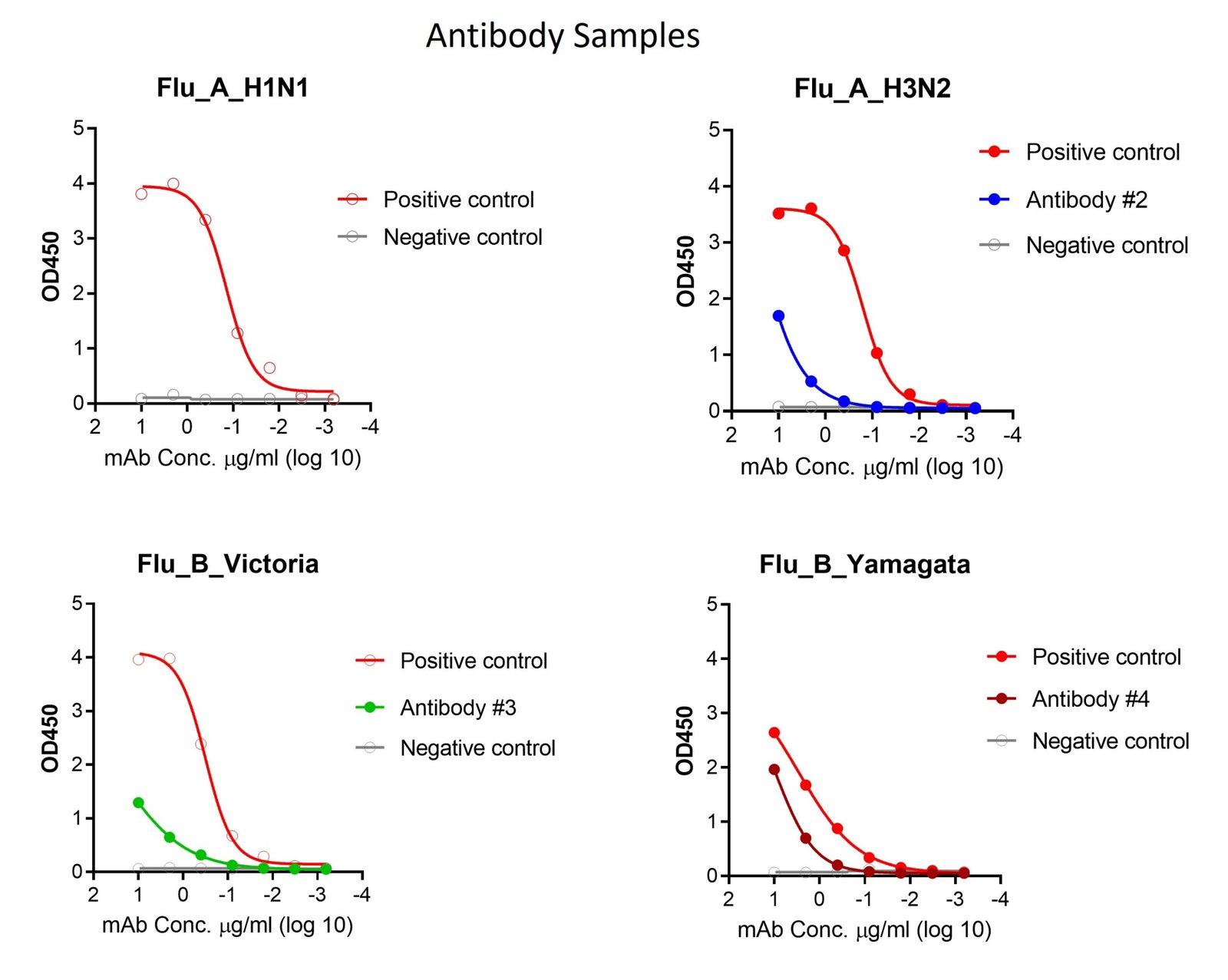
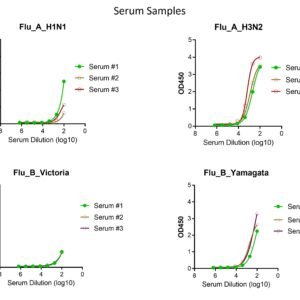
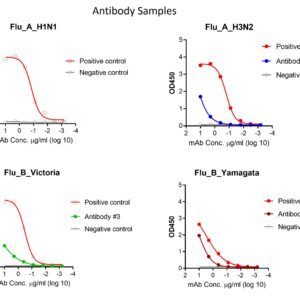
These kits were tested to bind to monoclonal antibodies and human sera, see figure.
Ordering:
B20S85-A-H1 for plate pre-coated with NP of Influenza A seasonal H1N1 (A/California/05/2009), recommended by WHO
B20S85-A-H3 for plate pre-coated with NP of Influenza A seasonal H3N2 (A/Darwin/9/2021)/(A/Darwin/6/2021), recommended by WHO
B20S85-B-V for plate pre-coated with NP of Influenza B seasonal Victoria Lineage (B/District Of Columbia/17/2019), recommended by WHO
B20S85-B-Y for plate pre-coated with NP of Influenza B seasonal Yamagata Lineage (B/Minnesota/15/2020), recommended by WHO
Biosafety Level:
BSL-1
Organism:
Influenza virus
Assay Principles:
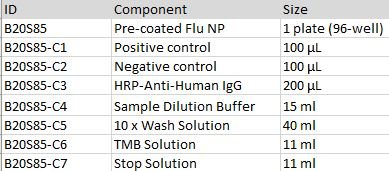
This kit is developed for a standard indirect-ELISA format, providing a rapid detection of anti-Influenza nucleoprotein (NP) human IgG in serum. The kit consists of high-bind detachable 96-well plate pre-coated with NP protein, Positive control, Negative Control, an HRP-Anti-Human IgG secondary antibody, TMB and dilution/wash/stop buffer.
Use protocol (4 simple steps):
- Detach wells from plate based on your samples and keep at room temperature. Dilute your samples, positive and negative controls in sample dilution buffer and add them to wells (0.1 ml/well). Incubate at 37°C for 1 hour or room temperature for 1.5 hour. Wash wells 4 times by wash solution (0.3 ml/well)
- Dilute the Secondary antibody HRP-Anti-Human IgG by wash solution and add to each well (0.1 ml/well). Incubate at room temperature for 0.5-1 hour. Avoid light by foil. Wash wells as mentioned.
- Add TMB to each well (0.1 ml/well) and incubate at room temperature for 10-30 minutes.
- Stop the substrate reaction by adding stop solution (0.1 ml/well). Absorbance (OD) is calculated as the absorbance at 450 nm. The OD Value reflects the amount of antibody bound.
Storage and shipping condition:
Shipment with room temperature. The unopened kit is stable for 6 months from the date of manufacture when stored at 2°C to 8°C. Avoid light. The shelf life is 60 days from the date of opening.