Lentivirus Packaging
Based on the human immunodeficiency virus-1 (HIV-1), lentivirus vector has been shown to deliver genes to neurons, lymphocytes and macrophages, cell types that previous retrovirus vectors could not be used. Lentiviral vector has also proven to be effective in transducing brain, liver, muscle, and retina in vivo without toxicity or immune responses. By integrating target gene into the host genome, lentivirus vector enables stable and long-term target gene expression—making them essential in gene therapy, cell therapy, and vaccine development.
We offer lentivirus packaging services with superior quality, together with VSV-G antibody kit to pre-screen VSV-G-specific antibody titer, which is essential for the success of lentiviral gene therapy. Besides traditional adhesive cell culture with serum (FBS) for the production of lentivirus, we offer advanced serum-free suspension culture to achieve high-titer lentivirus production up to 109 TU/mL with more purity.
Besides VSV-G, we provide a large panel of viral envelope you can choose from, such as CHIKV-E (Cat. No. H90Y16 containing a Luciferase reporter).
General Workflow
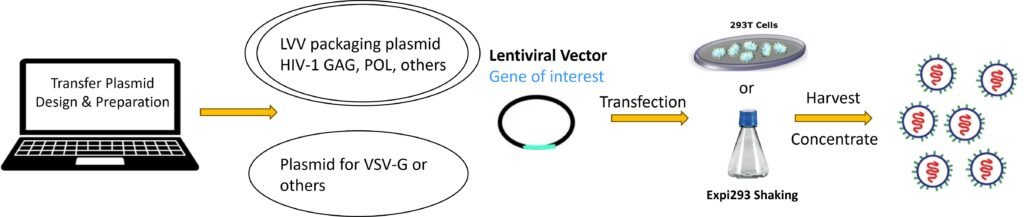
Service Details
Cat. No. | Scale | Application | Typical Titer | Volume | Price | Turnaround |
G21F73-1 | Supernatant | Cell culture | > 2E+6 TU/mL | 1 ml | $360 | 6-14 days |
G21F73-2 | Concentrated | > 1E+8 TU/mL | 200 ul | $400 | ||
G21F73-3 | 1 ml | $600 | ||||
G21F73-4 | > 1E+9 TU/mL | 1 ml | $1000 |
Note:
- The above table applies to VSV-G pseudotyped 2nd-generation lentivirus.
- By default, our Lentivirus Packaging services use 2nd generation lentiviral plasmids with VSV-G as the envelope plasmid.
- The volume and titer can be customized per request.
Quality Control
The default QC is by p24 ELISA (Cat. No. Q89H22). The calculated TU (Transduction units, also known as infectious units) is derived from the P24 titer equation and represents a predicted value, which may differ from actual functional titers.
The functional titering by qPCR is available upon request with extra cost.
If the transfer vector encodes a fluorescent protein, we would perform transduction test to detect corresponding fluorescence with extra cost. If the transfer vector encodes a drug-selectable marker, we would perform transduction test followed by corresponding drug selection with extra cost.
Transfer Plasmid
You provide a transfer plasmid containing gene of interest and LTR (selection marker is optional). (2nd or 3rd generation). See following table.
Element | Options |
Promoter | CMV, EF1α, U6, etc. |
Selection Marker | Puromycin, Hygromycin, Neomycin, Blasticidin, etc. |
Fluorescent Marker | GFP, Luciferase, mCherry, etc. |
Gene of Interest | De novo synthesis of the entire LTR to LTR cassette |
Or you can choose from one of ours (see below table). Or we help to design, for example we can modify ours to fit your interest. We can use P2A to control 2 genes under one promoter or 2 genes under 2 different promoters.
Name | Promoter | Selection | Cargo |
pLent-CMV-C-FH | CMV | Flag-His | |
pLent-CMV-C-HA | CMV | HA | |
pLent-CMV-N-Flag | CMV | Flag | |
pLent-CMV-N-Myc | CMV | Myc | |
pLent-CMV-N-HA | CMV | HA | |
pLent-CMV-C-GFP | CMV | GFP | |
pLent-CMV-C-RFP | CMV | RFP | |
pLent-CMV-N-GFP | CMV | GFP | |
pLent-CMV-N-RFP | CMV | RFP | |
pLent-CMV-P2A-COPGFP | CMV | GFP | |
pLent-CMV-P2A-PURO | CMV | Puromycin | Puro |
pLent-EF1a(CAR-T) | EF1α | ||
pLent-EF1a-FH-CMV-Puro | EF1α | Puromycin | Flag-His |
plent-EF1a-c-3flag-CMV-puro | EF1α | 3Flag | |
pLent-EF1a-FH-CMV-GFP-P2A-Puro | EF1α | Flag-His,GFP | |
pLent-EF1a-FH-CMV-copGFP-P2A-Puro | EF1α | Puromycin | Flag-His,GFP |
pLent-EF1a-Cre-CMV-copGFP-P2A-Puro | EF1α | Puromycin | GFP,Puro |
pLent-EF1a-3*Flag-CMV-GFP-P2A-Puro | EF1α | Puromycin | 3Flag,GFP |
pLent-EF1α-MF-CMV-GFP-P2A-Puro | EF1α | Puromycin | Myc-Flag,GFP |
pLent-EF1a-FH-CMV-RFP-P2A-Puro | EF1α,CMV | Puromycin | Flag-His,RFP |
pLent-EF1a-FH-CAG-mcherry-P2A-Puro | EF1α,CAG | Puromycin | Flag, His,mCherry |
pLent-EF1a-FH-CAG-GFP-P2A-Puro | EF1α,CAG | Puromycin | Flag, His,GFP |
pLent-EF1a-Luciferase-CMV-ECFP-P2A-Puro | EF1α,CMV | Puromycin | luciferase,ECFP |
pLent-EF1a-luciferase-CMV-copGFP-p2a-Puro | EF1α,CMV | Puromycin | luciferase,GFP |
pLent-EF1a-GFP-CMV | EF1α,CMV | GFP | |
plent-EF1a-puro-CMV-C-GFP | EF1α,CMV | GFP | |
pLent-EF1a-Puro-CMV | EF1α,CMV | Puro | |
pLent-EF1a-Blasticidin-CMV | EF1α,CMV | Blasticidin | |
pLent-EF1a-Puro-CMV-MF | EF1α,CMV | Myc-Flag | |
pLent-EF1A-PURO-CAG | EF1α,CAG | EF1α,CAG | |
pLent-SV40-Puro-CMV-MF | SV40,CMV | SV40,CMV | |
pLent-EF1a-circRNA-CMV-RFP-P2A-Puro | EF1α,CMV | Puromycin | circRNA,RFP |
pLent-EF1a-circRNA-CMV-GFP-P2A-Puro | EF1α,CMV | Puromycin | circRNA,GFP |
pLent-EF1a-puro-CMV-Luciferase | EF1α,CMV | Luciferase | |
pLent-EF1a-puro-CMV-Luciferase | EF1α,CMV | Luciferase | |
pLent-EF1a-puro-CD68-GFP | EF1α,CD68 | CD68 | |
pLent-EF1α-3flag-CMV-copGFP-P2A-Puro | EF1α,CMV | Puromycin | Flag,GFP |
pLent-EF1a-P2A-luciferase-CMV-copGFP-P2A-Puro | EF1α,CMV | Puromycin | luciferase,GFP |
pLent-EF1a-FH-CMV-RFP-P2A-Puro | EF1α,CMV | Puromycin | Flag, His, RFP |
pLent-EF1α-3flag-CMV-RFP-P2A-Puro | EF1α,CMV | Puromycin | Flag,RFP |
pLent-EF1a-FH-CMV-EBFP2-P2A-Puro | EF1α,CMV | Puromycin | Flag, His, EBFP2 |
pLent-EF1a-FH-CMV-tagBFP-P2A-Puro | EF1α,CMV | Puromycin | Flag, His, tgBFP |
pLent-EF1a-FH-CMV-Puro | EF1α,CMV | Puromycin | Flag, His |
pLent-EF1a-circRNA-CMV-RFP-P2A-puro | EF1α,CMV | Puromycin | circRNA,RFP |
pLenti-EF1a-FH-CMV-Blasticidin | EF1α,CMV | Blasticidin | Flag, His |
pLenti-EF1a-FH-CMV-luciferase-P2A-PURO | EF1α,CMV | Puromycin | Flag, His, Luciferase |
pLenti-EF1a-Luciferase-MIR-CMV-Puro | EF1α,CMV | Puromycin | Luciferase |
pLenti-EF1a-GFP-MIR-CMV-Puro | EF1α,CMV | Puromycin | GFP |
pLenti-EF1a-FH-CMV-copGFP-P2A-BSD | EF1α,CMV | Blasticidin | Flag, His,GFP |
pLent-EF1a-circRNA-cmv-copGFP-p2a-puro | EF1α,CMV | Puromycin | circRNA,GFP |
pLent-CAG-FH-CMV-copGFP-P2A-Puro | CAG,CMV | Puromycin | GFP |
pLent-EF1a-newcirc-CMV-copGFP-P2A-Puro | EF1α,CMV | Puromycin | GFP |
pLent-EF1a-newcirc-CMV-RFP-P2A-Puro | EF1α,CMV | Puromycin | RFP |
pLent-EF1a-luciferase-CMV-copGFP-P2A-puro | EF1α,CMV | Puromycin | luciferase,GFP |
pLent-EF1a-saCas9-CMV-copGFP-P2A-Puro | EF1α,CMV | Puromycin | spCas9,GFP |
pLent-TRE3G-FH-CMV-GFP-P2A-Puro | EF1α,CMV | Puromycin | Flag, His |
plent-ef1a-rfp-bsd-cmv-rtTA | EF1α,CMV | Blasticidin | rtTA |
pLent-TRE3G-3*FlaG-hPGK-rtta-puro | EF1α,CMV | Puromycin | Flag |
pLent-EF1a-puro-cmv-Luciferase-MIR | EF1α,CMV | Puromycin | Luciferase |
pLent-EF1a-Puro-CMV-GFP-MIR | EF1α,CMV | Puromycin | EF1a,CMV |
pLent-EF1A-GAG-CMV-PURO | EF1α,CMV | Puromycin | Puro |
pLent-TRE3G-zsGreen-mir30-shRNA-hPGK-rtTA | EF1α,CMV | zsGreen | |
pLent-TRE3G-zsGreen -hPGK-rtTA | EF1α,CMV | zsGreen | |
pLent-U6-shRNA-CMV-Puro | U6 | Puromycin | shRNA |
pLent-U6-shRNA-CMV-copGFP-p2a-Puro | u6 | Puromycin | shRNA, GFP |
pLent-U6-shRNA-CMV-RFP-p2a-Puro | U6 | Puromycin | shRNA, RFP |
pLent-U6-shRNA-CMV-RFP-p2a-Blasticidin | U6,CMV | Blasticidin | shRNA, GFP |
pLent-U6-shRNA-CMV-copGFP-P2A-Luciferase | U6,CMV | shRNA, GFP, Luciferase | |
plent-U6-shRNA-CMV-EBFP2-PURO | U6,CMV | Puromycin | shRNA,EBFP2 |
plent-U6-shRNA-CMV-EBFP2-BSD-NC | U6,CMV | Blasticidin | shRNA,EBFP2 |
pLent-U6-shRNA-CMV-copGFP-P2A-BSD | U6,CMV | Blasticidin | GFP |
pLent-U6-shRNA-CMV-luciferase-P2A-puro | U6,CMV | Puromycin | Luciferase |
pLent-U6-shRNA-CMV-Blasticidin | U6,CMV | Blasticidin | shRNA |
plent-2in1-spgRNA-EFFS-spcas9-p2a-copGFP | U6 | spCas9,GFP | |
pLent-U6-spgRNA-EFFS-spCas9-P2A-PURO | U6 | Puromycin | spCas9 |
pLent-U6-spgRNA-CMV-RFP-P2A-PURO | U6 | Puromycin | RFP |
pLent-EFFS-spCas9-P2A-BSD 52962 | U6 | Blasticidin | spCas9 |
pLent-spCas9-P2A-BSD-U6-sgRNA | U6 | Blasticidin | spCas9 |
pLent-CMV-spCas9-P2A-Puro | CMV | Puromycin | spCas9 |
pLent-U6-sagRNA-CMV-RFP | U6,CMV | Blasticidin | spgRNA,RFP |
plent-2in1 spgRNA-CMV-copGFP-P2A-Puro | CMV | Puromycin | spgRNA,GFP |
pLent-spgRNA-EF1a-spCas9-P2A-GFP-WPRE | EF1α | spCas9,GFP | |
pLent-gRNA-EFFS-spCAS9-EFFS-copGFP | U6 | spCas9,GFP | |
pLent-EF1a-P2A-Puro-CMV-copGFP | EF1α,CMV | Puromycin | GFP |
Experimental Validation & Reference lentivirus
See below reference lentivirus as example for experimental validation if your gene of interest has reporter or selection. If not, we have a large panel of antibody you can choose from to validate your gene of interest using flow cytometry.
Product Name | Catalog Number |
Lentivirus GFP-Luciferase Dual Reporter (Puromycin selection) | |
Lentivirus GFP Control (CMV Promoter) | |
Lentivirus Luciferase Control (CMV Promoter) |
Other Lentivirus products
Product Name | GOI | Reporter | Catalog |
Lentivirus Pseudotyped Chikungunya Virus E with Luciferase Reporter (LR2006_OPY1) | CHIKV-E | Luciferase | |
Lentivirus Pseudotyped Influenza A HPAI H5N1 virus HaNaM with Luciferase Reporter (clade 2.3.4.4b, 2022 strain) | Avian Flu-HA | Luciferase | |
Lentivirus Pseudotyped SARS-CoV (2003 strain) Spike with luciferase reporter | SARS-S | Luciferase | |
Lentivirus Pseudotyped SARS-CoV-2 (Wuhan Strain) Spike with luciferase reporter | SARS-CoV-2-S | Luciferase |
VSV-G-specific antibody pre-screening
The pre-determination of VSV-G-specific antibody level in recipient is essential for the success of lentiviral (LVV) therapy.
Human Anti-VSV-G Antibody IgG Titer Serologic Assay Kit (Cat. No. E9P59)
- ELISA Kit with VSV-G pre-coated
- Ready to use convenience
- Monoclonal antibody 8G5F11 to generate the reference curve
Or if you pre-test LVV in mice, you can choose:
Mouse Anti-VSV-G Antibody IgG Titer Serologic Assay Kit (Cat. No. D92H59)
- ELISA Kit with VSV-G pre-coated
- Ready to use convenience
