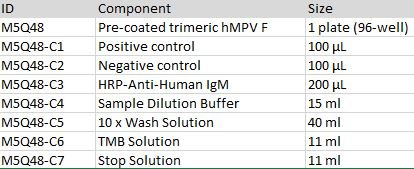
Human Anti-Metapneumovirus (hMPV) Fusion (F) Protein Antibody IgM Titer Serologic Assay Kit
Catalog Number:
M5Q48
Background:
The human metapneumovirus (hMPV) is a respiratory virus identified in 2001, even it has been circulating in humans for at least a half century. Almost all humans are infected with hMPV by the age of five. Two major genotypes, A and B, cause the similar clinical symptoms as closely resemble those seen with respiratory syncytial virus (RSV). The hMPV fusion (F) protein is a dominant antigen and a prominent vaccine target. A rapid and effective assay kit detecting the levels of anti-hMPV F in human serum can facilitate research on antibodies produced in response to hMPV infection or vaccine candidates.
Applications:
This kit is developed for serologic test for human IgM titer of Anti-hMPV (A1 or B1 subtype) F antibody in serum/plasma or purified human antibody (monoclonal & polyclonal) in vitro. It is for research use only.
Antibodies of IgM and IgA class are a sign of an active infection (primary infection and reactivation) and disappear during convalescence. In some cases they can persist for several months.
Description:
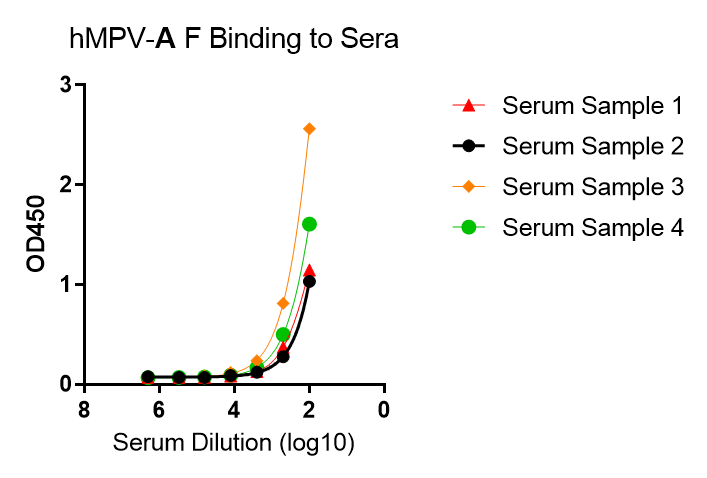
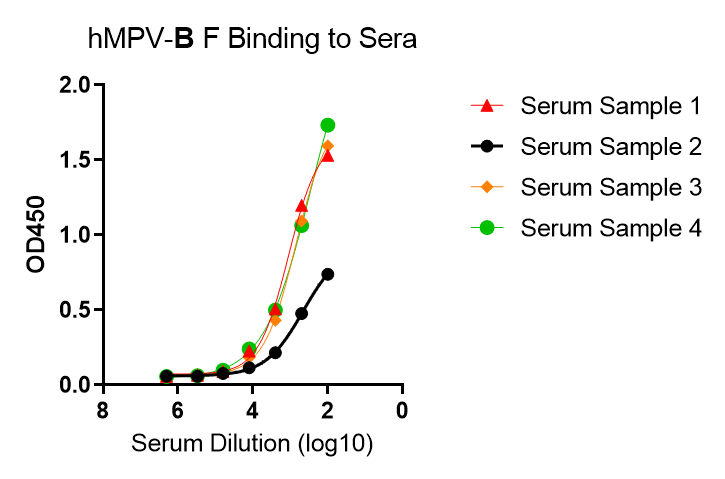
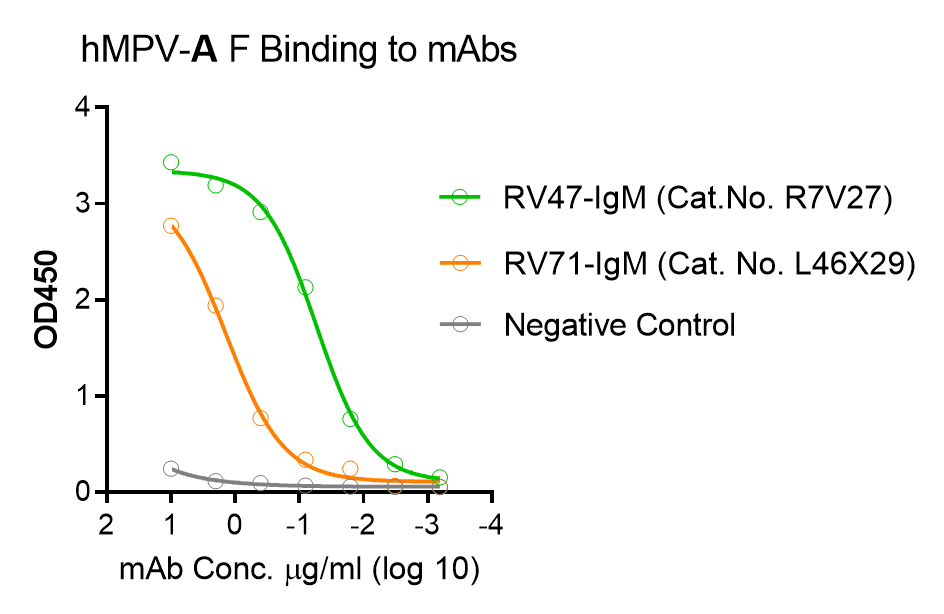
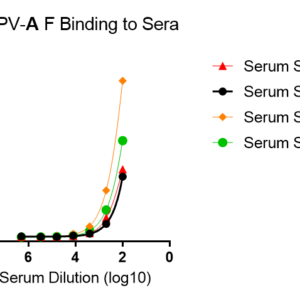
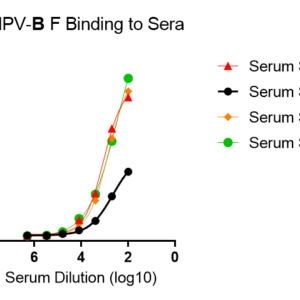
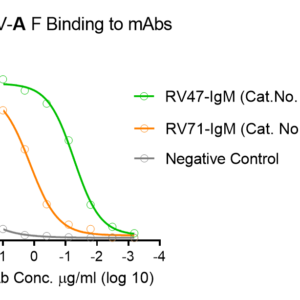
These kits were tested to bind to monoclonal antibodies in IgM format (clone RV47, Cat. No. R7V27 or clone RV71, Cat. No. L46X29) or human sera, see figures.
Ordering:
M5Q48-A for plate pre-coated with prefusion-stabilized trimeric fusion (F) protein of hMPV isolate NL/1/00 A1.
M5Q48-B for plate pre-coated with prefusion-stabilized trimeric fusion (F) protein of hMPV isolate NL/1/99 B1.
Biosafety Level:
BSL-1
Organism:
Human metapneumovirus (hMPV)
Assay Principles:
This kit is developed for a standard indirect-ELISA format, providing a rapid detection of anti-hMPV fusion (F) protein human IgM in serum. The kit consists of high-bind detachable 96-well plate pre-coated with trimeric F protein, Positive control, Negative Control, an HRP-Anti-Human IgM secondary antibody, TMB and dilution/wash/stop buffer.
Use protocol (4 simple steps):
- Detach wells from plate based on your samples and keep at room temperature. Dilute your samples, positive and negative controls in sample dilution buffer and add them to wells (0.1 ml/well). Incubate at 37°C for 1 hour or room temperature for 1.5 hour. Wash wells 4 times by wash solution (0.3 ml/well)
- Dilute the Secondary antibody HRP-Anti-Human IgM by wash solution and add to each well (0.1 ml/well). Incubate at room temperature for 0.5-1 hour. Avoid light by foil. Wash wells as mentioned.
- Add TMB to each well (0.1 ml/well) and incubate at room temperature for 10-30 minutes.
- Stop the substrate reaction by adding stop solution (0.1 ml/well). Absorbance (OD) is calculated as the absorbance at 450 nm. The OD Value reflects the amount of antibody bound.
Storage and shipping condition:
Shipment with room temperature. The unopened kit is stable for 6 months from the date of manufacture when stored at 2°C to 8°C. Avoid light. The shelf life is 60 days from the date of opening.
Recommendations:
Customers who intend to use this kit to generate quantitative curves are suggested to consider the monoclonal antibodies to generate the reference curve: Recombinant Human Anti-hMPV Fusion IgM Antibody (Cat. No. R7V27) or Recombinant Human Anti-RSV and HMPV Cross-reactive F IgM Antibody (Cat. No. L46X29).