Respiratory Syncytial Virus (RSV) Surrogate Virus Neutralization Assay Kit (Fusion, F)
Catalog Number:
S93V69
Background:
Respiratory syncytial virus (RSV) annually causes approximately 3 million acute lower respiratory infections in children and is typified by recurrent infections in infants. Two major RSV subtypes, A and B, circulate with 1-2 epidemic periods per year. The RSV fusion (F) glycoprotein is a dominant antigen and a prominent vaccine target. A rapid and effective assay kit detecting the neutralizing levels of anti-RSV F from both subtypes A and B in human serum can facilitate research on antibodies produced in response to RSV infection or vaccine candidates.
Applications:
This kit is developed to measure circulating neutralizing antibodies or other molecules against RSV that block the interaction between viral fusion F with its receptor. The assay detects any antibodies in serum/plasma or purified antibody (monoclonal & polyclonal) or other molecules that inhibit F binding to its receptor in vitro. The test is both species and isotype independent. It is for research use only.
Description:
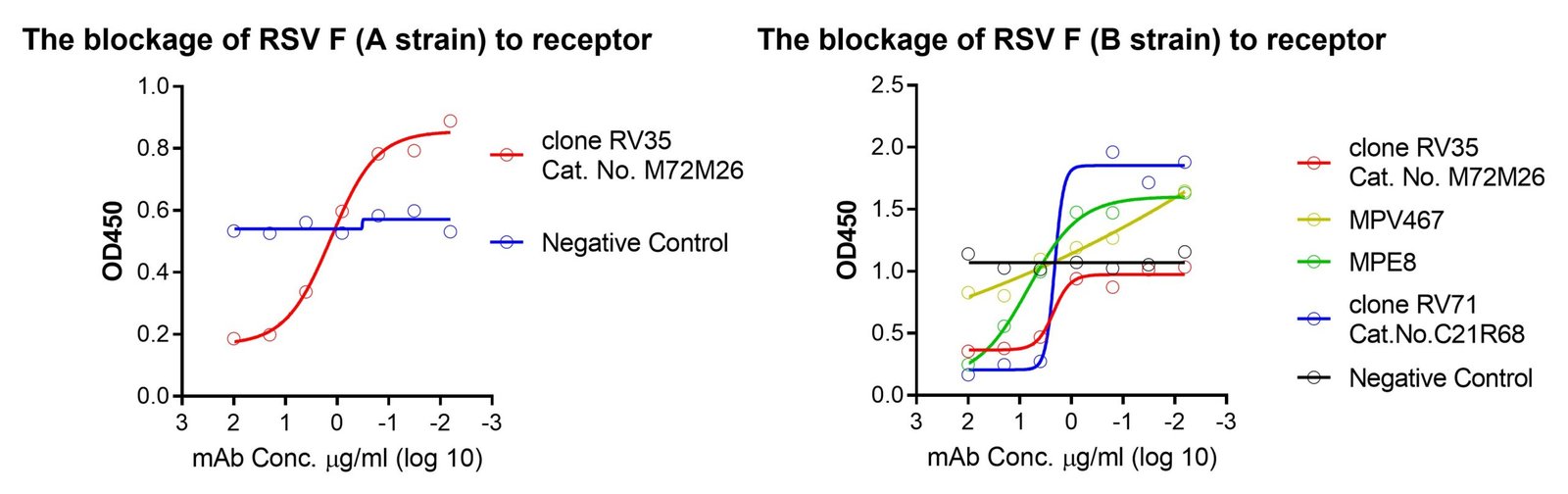
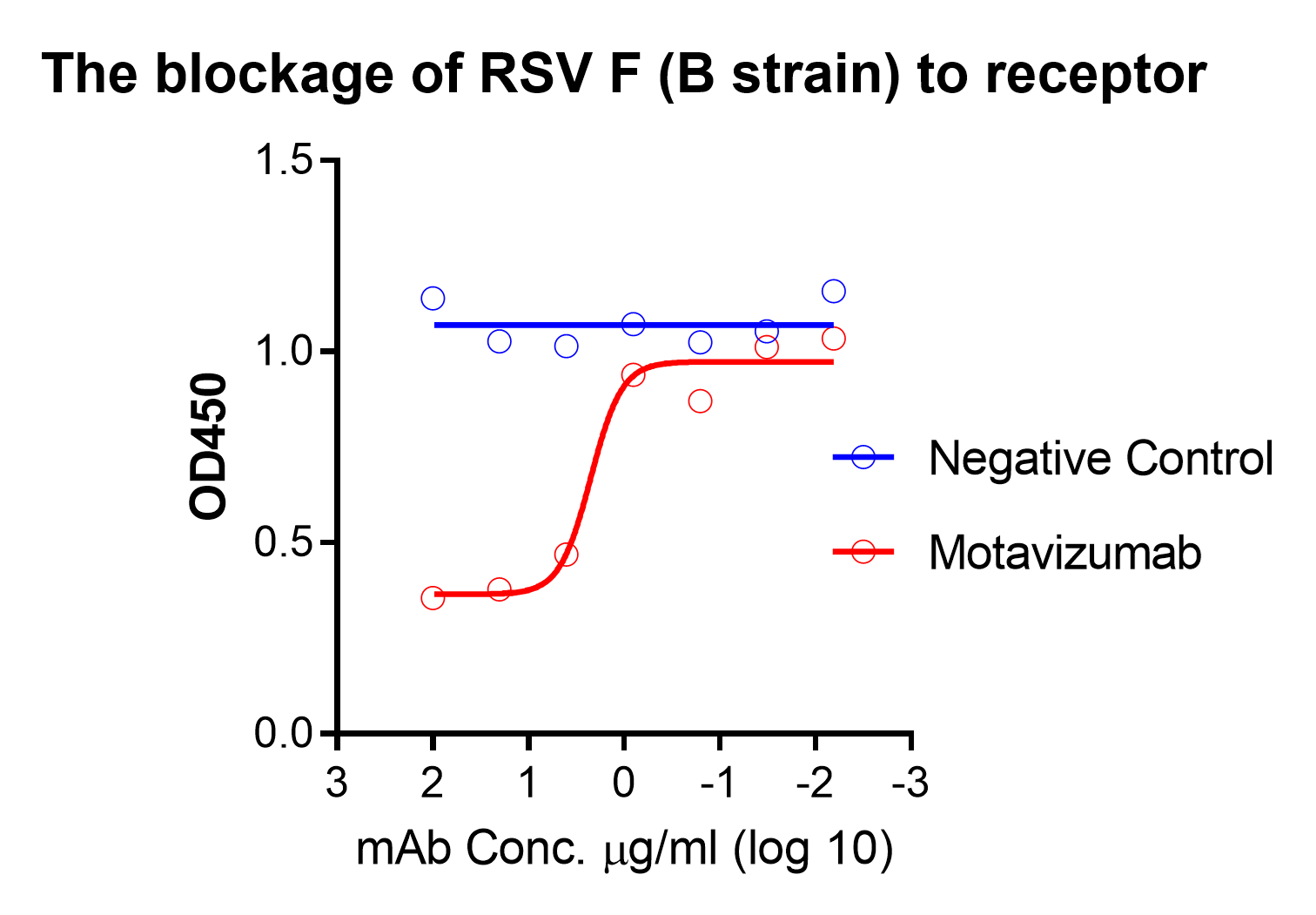
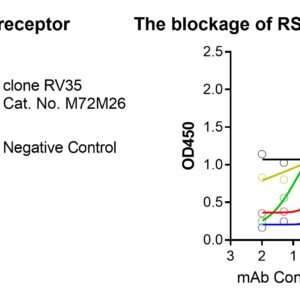
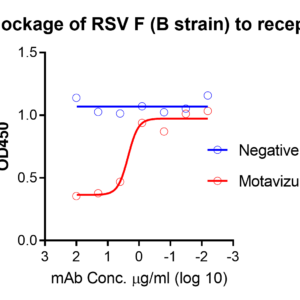
These kits were validated by multiple known monoclonal neutralizing antibodies – MPE8, MPV467, clone RV71 (Anti-RSV and HMPV Cross-reactive F Monoclonal Antibody, Cat. No. C21R68) or clone RV35 (Cat. No. M72M26, Motavizumab similar). The binding between F and receptor is blocked by these neutralizing antibodies in a dose-dependent manner, see figures.
Ordering:
S93V69-A for neutralization test of RSV subtype A strain A2.
S93V69-B for neutralization test of RSV subtype B strain 18537.
Biosafety Level:
BSL-1
Organism:
Respiratory syncytial virus (RSV)
Assay Principles (Take neutralizing antibody as example):
This kit is developed for a blocking ELISA format to mimics the virus neutralization process in vitro. The samples or controls are pre-incubated with RSV F, and then added to the capture plate which is pre-coated with receptor(s). The unbound F as well as any F bound to non-neutralizing antibody will be captured on the plate, while the neutralizing antibody-F complexes remain in the supernatant and get removed during washing. After detecting and reading at 450 nm, the absorbance of the sample is inversely dependent on the titer of the anti-RSV F neutralizing antibodies.
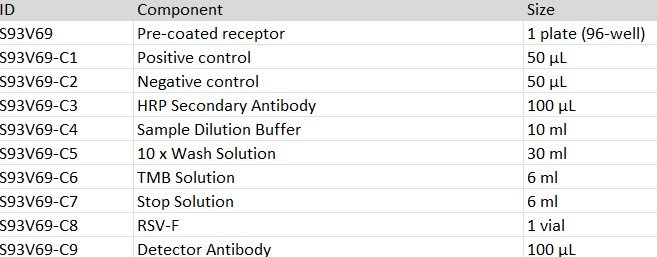
The kit consists of 96-well plate with receptor pre-coated, Positive control, Negative Control, RSV-F, detector antibody, HRP labeled secondary antibody, TMB and dilution/wash/stop buffer.
Brief protocol (detailed protocol is provided with the product):
- Dilute RSV-F protein using provided sample dilution buffer based on the amount of your sample and mix with your sample or provided controls. Set few dilution of your sample. Incubate at room temperature for 1 hour.
- Transfer above mixture to provided detachable plate which receptor was pre-coated (50 µl/well). Incubate at room temperature for 1 hour.
- Wash wells 4 times using wash solution (0.2 ml).
- Add detector antibody (50 µl/well). Incubate at room temperature for 1 hour. Wash wells as mentioned.
- Add HRP labeled secondary antibody to each well (50 µl /well). Incubate at room temperature for 1 hour. Avoid light by foil. Wash wells as mentioned.
- Add TMB to each well (50 µl /well) and incubate at room temperature for 10-30 minutes.
- Stop the substrate reaction by adding stop solution (50 µl /well). Absorbance (OD) is calculated as the absorbance at 450 nm. The OD Value inversely reflects the amount of neutralizing antibody bound.
Storage and shipping condition:
Shipment with room temperature. The unopened kit is stable for 6 months from the date of manufacture when stored at 2°C to 8°C. Avoid light. The shelf life is 60 days from the date of opening.
Recommendations:
Customers who intend to use this kit to generate quantitative curves are suggested to consider the monoclonal antibodies to generate the reference curve: Anti-RSV Fusion Human Monoclonal Antibody (Cat. No. M72M26).